7.2 Protein Synthesis and Denaturation
Protein Synthesis
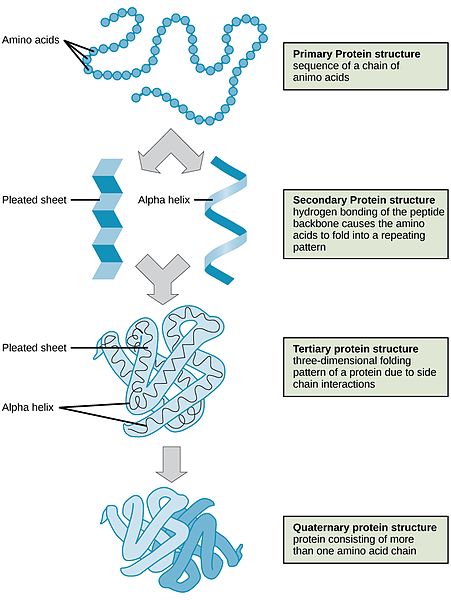
Protein synthesis refers to a process that takes place in the cells when proteins are created from amino acids. As there are only 20 amino acids that your cells use to create thousands of different proteins, protein synthesis is very complex. Protein synthesis has four distinct steps or levels of structure as the individual amino acids are converted into functional proteins.
- Primary: The first level is the one-dimensional sequence of amino acids that are held together by peptide bonds to form a polypeptide.
- Secondary: The second level of protein structure is dependent on the chemical interactions between amino acids, which cause the protein to fold into a specific shape, such as a helix (like a coiled spring) or sheet.
- Tertiary: The third level of protein structure is three-dimensional. As the different side chains of amino acids chemically interact, they either repel or attract each other, resulting in the folded structure. Thus, the specific sequence of amino acids in a protein directs the protein to fold into a specific, organized shape. At this point, some proteins are functional; however, most require a fourth step.
- Quaternary: The fourth level of structure is achieved when protein fragments called peptides combine with each other or with a vitamin or mineral to make one larger functional protein. You can think of this as “activating” the protein so it is functional. The protein hemoglobin is an example of a protein that has quaternary structure. It is composed of four peptides that bond together to form a functional oxygen carrier.
Figure 7.4 Steps of Protein Synthesis
Proteins have different shapes and molecular weights. Protein shape is critical to its function. Changes in temperature, pH, and exposure to chemicals may lead to permanent changes in the shape of the protein leading to a loss of function or denaturation.
Denaturation
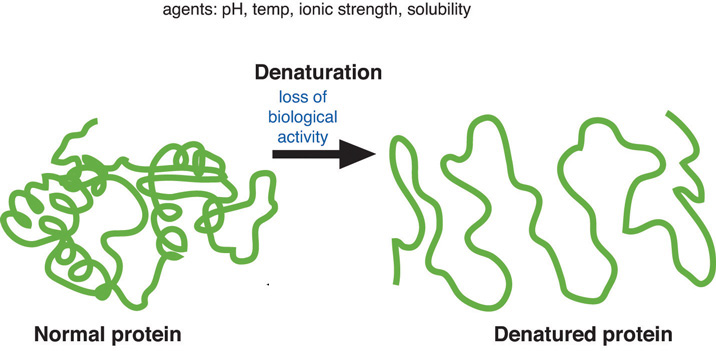
When a cake is baked, the proteins are denatured. Denaturation refers to the physical changes that take place in a protein exposed to abnormal conditions in the environment. Heat, acid, high salt concentrations, alcohol, and mechanical agitation can cause proteins to denature. When a protein denatures, its complicated folded structure unravels, and it becomes just a long strand of amino acids again. Weak chemical forces that hold tertiary and secondary protein structures together are broken when a protein is exposed to unnatural conditions. Because proteins’ function is dependent on their shape, denatured proteins are no longer functional. During cooking the applied heat causes proteins to vibrate. This destroys the weak bonds holding proteins in their complex shape (though this does not happen to the stronger peptide bonds). The unraveled protein strands then stick together, forming an aggregate (or network).
Figure 7.5 shows how the shape of a protein changes when it is denatured. When a protein is exposed to a different environment, such as increased temperature, it uncoils and unfolds into a single strand of amino acids.
Figure 7.5 Protein Denaturation
Media Attributions
- Steps of Protein Synthesis © OpenStax is licensed under a CC BY (Attribution) license
- Denaturation of Proteins © University of Hawaii is licensed under a CC BY-NC-SA (Attribution NonCommercial ShareAlike) license
